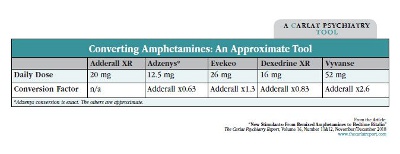
Adderall Xr To Ir Conversion, When it comes to managing ADHD symptoms, Adderall is a commonly prescribed medication. Adderall is available in two formulations: Adderall XR (extended-release) and Adderall IR (immediate-release). Each has its unique benefits and considerations, and sometimes patients may need to switch from one to the other. Understanding the conversion process between Adderall XR and Adderall IR is crucial for maintaining effective symptom management.
What is Adderall?
Adderall is a stimulant medication containing amphetamine and dextroamphetamine. It’s used primarily to treat Attention Deficit Hyperactivity Disorder (ADHD) and narcolepsy. The medication works by increasing the levels of certain neurotransmitters in the brain, helping to improve focus, attention, and impulse control.
Adderall XR vs. Adderall IR
- Adderall XR (Extended-Release):
- Duration: Designed to last up to 12 hours.
- Release Mechanism: Contains beads that release the medication at different times, providing a steady release throughout the day.
- Dosing: Usually taken once a day in the morning.
- Advantages: Convenient for all-day symptom control, reducing the need for multiple doses.
- Adderall IR (Immediate-Release):
- Duration: Typically lasts 4-6 hours.
- Release Mechanism: Provides an immediate boost of medication, with a quick onset of action.
- Dosing: Usually taken multiple times a day (e.g., morning and early afternoon).
- Advantages: Allows for more flexible dosing and fine-tuning of medication levels throughout the day.
Why Convert from XR to IR?
There are several reasons why a patient might need to switch from Adderall XR to Adderall IR:
- Variable Schedule: Patients with variable daily schedules may benefit from the flexibility of IR dosing.
- Side Effects: Some individuals may experience fewer side effects with IR formulations.
- Dosing Adjustments: IR allows for easier adjustments in dosing, which can be useful during periods of increased or decreased need.
- Insurance and Cost: In some cases, insurance coverage or medication costs might necessitate a switch.
How to Convert from Adderall XR to IR
Conversion from Adderall XR to Adderall IR should always be done under the guidance of a healthcare provider. Here’s a general outline of the process:
- Initial Assessment: The healthcare provider will evaluate the current dose of Adderall XR and the patient’s response to it.
- Dose Calculation: Generally, the total daily dose of Adderall XR is divided into two or three doses of Adderall IR. For example, if a patient is taking 20 mg of Adderall XR once daily, the equivalent IR dose might be 10 mg taken twice a day (morning and early afternoon).
- Monitoring: The patient will need to closely monitor their symptoms and any side effects during the transition. This may involve keeping a symptom diary and having regular check-ins with the healthcare provider.
- Adjustments: Based on the patient’s response, the healthcare provider may make further adjustments to the IR dosing schedule or amounts to optimize symptom control and minimize side effects.
Tips for a Smooth Transition
- Communicate: Keep an open line of communication with your healthcare provider. Report any changes in symptoms or side effects promptly.
- Consistency: Take your medication at the same times each day to maintain consistent levels in your bloodstream.
- Support System: Inform family members or caregivers about the change in medication so they can provide support and monitor for any changes in behavior or symptoms.
- Patience: Allow some time for your body to adjust to the new dosing regimen. It may take a few weeks to find the optimal dose and schedule.
Conclusion
Switching from Adderall XR to Adderall IR can offer flexibility and potentially better symptom control for some individuals. However, it’s a process that requires careful planning and monitoring. Always work closely with your healthcare provider to ensure a safe and effective transition. With the right approach, you can manage your ADHD symptoms effectively and improve your overall quality of life.